Water in the chest – new findings on pleural effusion
Lung cancer patients are particularly susceptible to malignant pleural effusion, when fluid collects in the space between the lungs and the chest wall. Researchers at the Helmholtz Zentrum München, in partnership with the German Center for Lung Research (DZL), have discovered a novel mechanism that causes this to happen. Their study, published in ‘Nature Communications’, now refines the mechanistic picture.
Malignant pulmonary effusion (MPE) frequently occurs in patients with metastatic breast or lung cancer. It involves a build-up of excess fluid in the pleural cavity, the area between the lungs and the chest wall, with accompanying malignant cells. The lung is surrounded by fluid, which can cause shortness of breath and chest pain, for example, and may even prove fatal.
“The cause is still not fully understood, which makes the search for suitable therapies more difficult,” explains Professor Georgios Stathopoulos, research group leader at the Institute of Lung Biology (ILBD) and the Comprehensive Pneumology Center (CPC) at Helmholtz Zentrum München. “However, we’ve now made significant progress in that direction.”
In their recent work, the team built on findings which they had also published in ‘Nature Communications’ in May 2017. “At the time, we were able to show that pleural effusion is triggered by cancer cells with a malignant mutation in the KRAS gene*,” says Dr. Antonia Marazioti, lead author of the paper and scientist at the Molecular Respiratory Carcinogenesis Research Group in the medical department of the University of Patras, which is also led by Georgios Stathopoulos in close connection to his ILBD/CPC work at the Helmholtz Zentrum München.
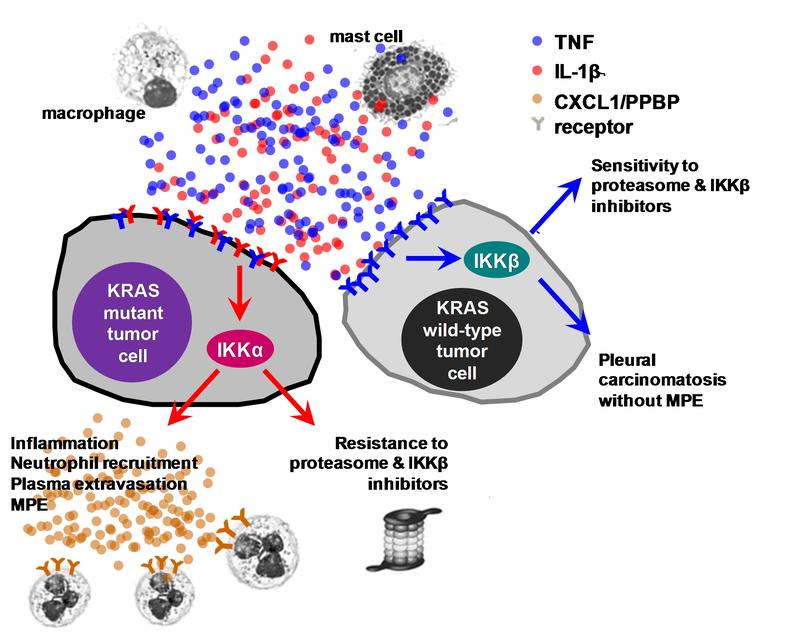
The authors have now been able to expand on this knowledge. “Our experiments show that inflammatory messengers of the immune system − notably interleukin-1β − activate a signaling pathway in mutated cancer cells, which in the long term can lead to pleural effusion,” Stathopoulos explains. The molecule IKKα** plays a key role in the signaling pathway in that it, in turn, releases other messengers (CXCL1), resulting in a strong inflammatory response (see illustration). “Consequently, those cells migrate to the pleural cavity via the spleen, where they cause effusion,” explains the lung expert.
Double-pronged inhibition is better
To determine whether the findings might prove relevant to future treatment strategies, the researchers suppressed the newly discovered signaling pathway in the experimental model from two sides. In their double-pronged approach, they used both an inhibitor of KRAS and an inhibitor of IKKα. “In fact, this dual strategy significantly reduced both the incidence and the progression of MPE,” Stathopoulos reports. Resistance to a single treatment was also reduced.
“Nearly two thirds of all MPEs are the result of lung cancer. Given the large number of people who still smoke, appropriate therapies are urgently needed,” Georgios Stathopoulos says. “Our findings suggest that drugs could become a therapeutic option for suppressing the mechanism we’ve discovered. We plan to investigate this line of enquiry in greater depth in the future and to further confirm the results in the translational approach with lung cancer patients in collaboration with the Asklepiosklinik in Gauting.”
Further Information
* KRAS is known to play a key role in the growth of various malignancies.
** IKKα stands for inhibitor of nuclear factor-κΒ kinase α.
Background:
Co-authors Malamati Vreka and Georgia A. Giotopoulou are doctoral students at the CPC Research School and participants in the Helmholtz Graduate School Environmental Health doctoral education program, or HELENA for short.
Original Publication:
Marazioti, A. et al. (2018): Myeloid-derived Interleukin-1β drives oncogenic KRAS-NF-κΒ addiction in malignant pleural effusion. Nature Communications, DOI: 10.1038/s41467-018-03051-z
The Helmholtz Zentrum München, the German Research Center for Environmental Health, pursues the goal of developing personalized medical approaches for the prevention and therapy of major common diseases such as diabetes and lung diseases. To achieve this, it investigates the interaction of genetics, environmental factors and lifestyle. The Helmholtz Zentrum München is headquartered in Neuherberg in the north of Munich and has about 2,300 staff members. It is a member of the Helmholtz Association, a community of 18 scientific-technical and medical-biological research centers with a total of about 37,000 staff members. www.helmholtz-muenchen.de/en
The Comprehensive Pneumology Center (CPC) is a joint research project of the Helmholtz Zentrum München, the Ludwig-Maximilians-Universität Clinic Complex and the Asklepios Fachkliniken München-Gauting. The CPC's objective is to conduct research on chronic lung diseases in order to develop new diagnosis and therapy strategies. The CPC maintains a focus on experimental pneumology with the investigation of cellular, molecular and immunological mechanisms involved in lung diseases. The CPC is a site of the Deutsches Zentrum für Lungenforschung (DZL). www.helmholtz-muenchen.de/ilbd
The German Center for Lung Research (DZL) pools German expertise in the field of pulmonology research and clinical pulmonology. The association’s head office is in Giessen. The aim of the DZL is to find answers to open questions in research into lung diseases by adopting an innovative, integrated approach and thus to make a sizeable contribution to improving the prevention, diagnosis and individualized treatment of lung disease and to ensure optimum patient care. www.dzl.de/index.php/en
Contact for the media:
Department of Communication, Helmholtz Zentrum München - German Research Center for Environmental Health, Ingolstädter Landstr. 1, 85764 Neuherberg - Tel. +49 89 3187 2238 - Fax: +49 89 3187 3324 - E-mail: presse@helmholtz-muenchen.de
Scientific Contact:
Prof. Dr. Georgios Stathopoulos, Helmholtz Zentrum München - German Research Center for Environmental Health, Comprehensive Pneumology Center, Max-Lebsche-Platz 31, 81377 München, Germany - Tel. +49 89 3187 4846 - E-mail: stathopoulos@helmholtz-muenchen.de
Ähnliche Pressemitteilungen im idw