The way of making memories
Human beings live and learn and this usually happens through experience and environmental interactions. But how does the brain translate information from the outside world into something we subsequently remember? An international team of researchers in Germany, Sweden and Switzerland working in the Human Brain Project ( www.humanbrainproject.eu ) have now zoomed in on the neuronal circuits in the striatum, a brain structure involved in memory, behavior and reward learning. The findings, published in the PLOS Computational Biology Journal, increase our knowledge of the basic functioning of the nervous system and its ability to learn and adapt to changing conditions.
We all know that experience: We hear a tune and it somehow leaves its mark so that we recognize it when we hear it again, even decades later. We look at a van Gogh painting once and it remains impressed in our memory for all our life. But how can something so evanescent as a melody become part of the brain and lead to the formation of memories that shape our behavior?
Information processing in the brain occurs within neuronal circuits that are interconnected by synapses. Each modification of these synapses has an impact on how we remember things or react to certain stimuli. One way that neuronal circuits are modified is through the process of synaptic plasticity, where certain synapses are either strengthened or weakened over time in response to neural activity. By analyzing the networks of biochemical reactions that underlie synaptic modifications, scientists in Heidelberg, Lausanne, Juelich and Stockholm have been able to gain new insights into the mechanisms of plasticity.
“Simulations of plasticity mechanisms are crucial if we want to enhance our understanding of how higher level phenomena, such as learning and memory formation, arise from computations at the molecular level,” says Jeanette Hellgren Kotaleski from the Royal Institute of Technology in Stockholm, one of the co-leaders of the study.
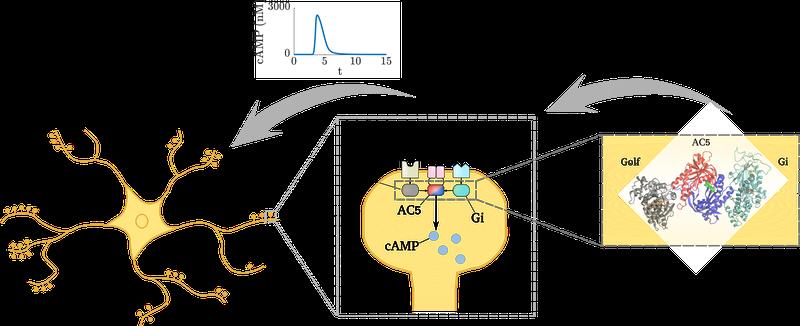
In neurons, external and internal information processing occurs through synaptic signal transduction networks which determine synaptic plasticity. Sometimes even single molecules – often enzymes, proteins that greatly accelerate or catalyze specific chemical reactions – are able to realize computational abilities within these networks. One such case is the family of mammalian adenylyl cyclase enzymes (ACs), that can translate extracellular signals into the intracellular molecule cAMP, one of the main cellular second messenger signaling molecules. “I have been fascinated by these enzymes for a long time,” says Paolo Carloni from Forschungszentrum Juelich, “as what is really important here is not how fast the catalyzed reaction is but rather how Nature exerts a tight control on these chemical machines: specific accessory proteins, by exquisitely targeting AC enzymes, kick the chemical reactions off, others block them. Our work provides a significant step towards understanding what we can call “molecular recognition” of these AC proteins, based on which neurons can control with astonishing precision and fidelity the speed of AC’s catalyzed reaction. This in turn activates subsequent downstream processes essential for neuronal function.”
The brain expresses nine membrane-bound AC variants, and AC5 is the dominant form in the striatum. During reward learning, cAMP production is crucial for strengthening the synapses from cortical neurons onto the striatal principal neurons, and its formation is dependent on several neuromodulatory systems such as dopamine and acetylcholine. “For this study, it was necessary to bring the different computational expertise of scientists in four laboratories together to work on a multiscale simulation approach to constructing a kinetic model of the AC5-dependent signaling system,” says Rebecca Wade who led the study at the Heidelberg Institute for Theoretical Studies (HITS). “From this model, we could find out how AC5 can detect particular combinations of simultaneous changes in neuromodulatory signals which result in a synergistic cAMP production.“
The results provided in this paper include contributions from the research groups of Rebecca Wade from the Heidelberg Institute for Theoretical Studies (HITS), Ursula Roethlisberger from the École Polytechnique Fédérale de Lausanne, Paolo Carloni from the Forschungszentrum Juelich and Jeanette Hellgren Kotaleski from the Royal Institute of Technology, Stockholm. The project has received funding from the Human Brain Project.
Wissenschaftlicher Ansprechpartner:
Prof. Dr. Rebecca Wade
Molecular and Cellular Modeling group
Heidelberg Institute for Theoretical Studies (HITS)
Phone: +49 6221 533 245
rebecca.wade@h-its.org
Prof. Dr. Paolo Carloni
Institute of Advanced Simulation
Forschungszentrum Jülich
Phone: +49 2461 61 8941
p.carloni@fz-juelich.de
Prof. Dr. Jeannette Hellgren Kotaleski
KTH Royal Institute of Technology, Stockholm
Phone: +46 8 524 871 80
jeannette.hellgren@ki.se
Originalpublikation:
Bruce NJ, Narzi D, Trpevski D, van Keulen SC, Nair AG, et al. (2019) Regulation of adenylyl cyclase 5 in striatal neurons confers the ability to detect coincident neuromodulatory signals. PLOS Computational Biology 15(10): e1007382. https://doi.org/10.1371/journal.pcbi.1007382
Weitere Informationen:
https://www.h-its.org/2019/11/07/ac5-en/ HITS press release
Ähnliche Pressemitteilungen im idw