A disease trigger for pancreatitis has been identified
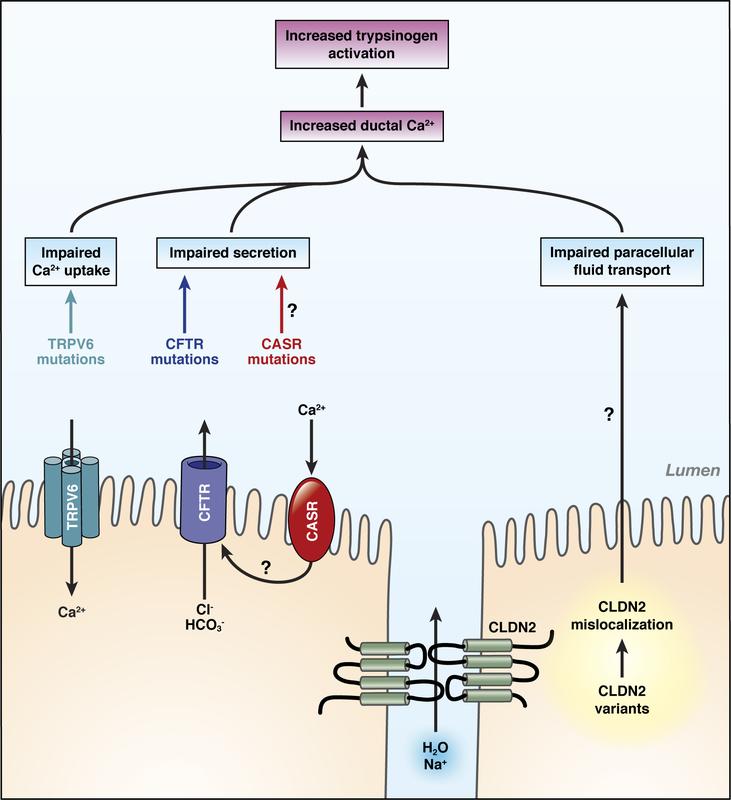
One factor contributing to the development of pancreatitis lies in mutations within a cell membrane ion channel that is characterized by its specific permeability for calcium ions. This groundbreaking discovery was made by a research team at Technical University of Munich (TUM), in cooperation with other groups from Germany, Japan and France. These insights could lead to the development of new therapeutic agents for treating and preventing chronic pancreatitis.
Patients suffering from chronic pancreatitis experience an either recurring or permanent inflammation of their pancreas. “In many cases, people develop this disease because they are drinking too much alcohol or they are smoking too much. Certain medication or high levels of lipids or calcium in a patient’s blood can be another cause of pancreatitis,” explained Heiko Witt, one of the two heads of the study and Professor for Pediatric Nutritional Medicine at the Else Kröner-Fresenius-Zentrum (EKFZ) at TUM.
The main focus of previous research was on the so-called acinus cells of the pancreas; these are responsible for creating digestive enzymes. Many patients suffering from genetically caused pancreatitis show mutations in digestive enzymes or in molecules inhibiting the enzymes’ effectiveness.
Calcium channel disorder leads to pancreatitis
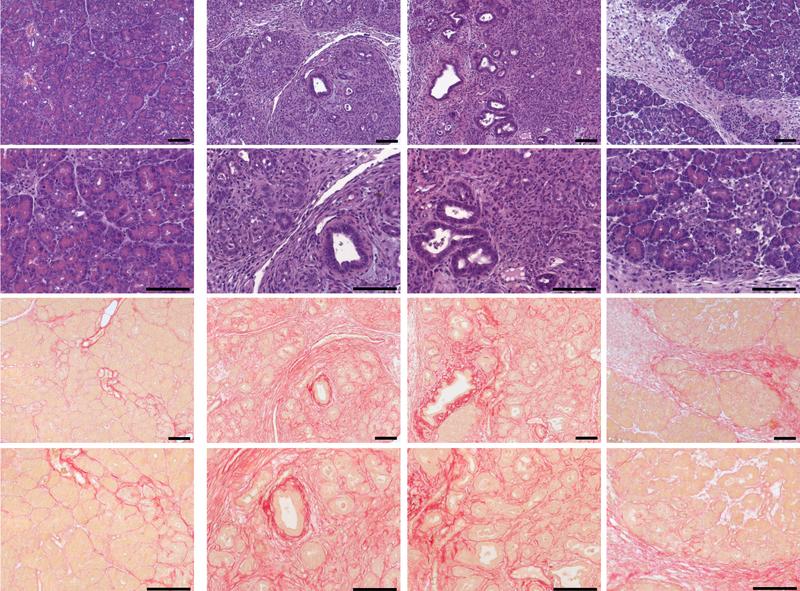
In the course of the current study, that was performed with European and Japanese patients suffering from pancreatitis not associated with alcohol consumption, the researchers discovered that genetic defects which were heavily limiting the functionality of calcium channel TRPV6 caused early-onset chronic pancreatitis.
“A substantial TRPV6 defect is a globally occurring and serious risk factor for developing chronic pancreatitis,“ said Professor Witt and added: “By identifying alterations of the calcium channel, we are now also considering duct cells as part of the concept covering the origins of this disease.” Duct cells serve as coating in the channels that transport digestive enzymes from the point of origin into the intestines.
Using a mouse model, the scientists were able to show that the absence of the corresponding gene would, in most cases, lead to inflammation and the occurrence of fibrotic changes in the pancreas, which is typical for chronic inflammation.
This new discovery offers opportunities for pharmacologic therapy approaches
The discovery that a calcium channel malfunction can contribute to the development of pancreatitis offers a new course of action for therapeutic intervention. Furthermore, the research findings will become part of the standard diagnostics for genetically caused pancreatitis.
These insights also pave the way for a new area of research covering the investigation of causes of pancreatitis – no longer focused on acinus cells and digestive enzymes but rather on duct cells and channels as well as calcium metabolism.
The identification of mutations in a calcium channel as a (contributing) cause of disease leads to new factors – other calcium channels and proteins that are relevant for calcium metabolism – moving into the scientific focus. “Currently we are investigating these genes for genetic alterations and we have a European patient collective comprising 1100 people with pancreatitis to gather our data,” reported Professor Witt.
He further explained: “Deciphering the genetic backgrounds of pancreatitis will substantially affect our understanding of these types of genetically caused pancreatitis, as well as our understanding of alcohol-related pancreatitis. It will enable new research approaches which may lead to new treatment possibilities in the future.”
Wissenschaftlicher Ansprechpartner:
Prof. Dr. Heiko Witt
Technical University of Munich
Professor of Paediatric Nutritional Medicine
Else Kröner-Fresenius Zentrum (EKFZ)
phone: +49.8161.71.2466
Heiko.Witt@tum.de
Originalpublikation:
Masamune A., Kotani H., Sörgel F.L., [...] , Witt H., Shimosegawa T.: Variants That Affect Function of Calcium Channel TRPV6 Are Associated With Early-Onset Chronic Pancreatitis. Gastroenterology. 2020 Jan 10. pii: S0016-5085(20)30017-2. doi: 10.1053/j.gastro.2020.01.005.
Weitere Informationen:
https://www.ncbi.nlm.nih.gov/pubmed/31930989 (publication)
https://www.pem.wzw.tum.de/en/home/ (Homepage: Paediatric Nutritional Medicine)
https://www.professoren.tum.de/en/witt-heiko/ (Prof. Witt)
Die semantisch ähnlichsten Pressemitteilungen im idw