Researchers classify disease severity of patients with Li-Fraumeni syndrome for the first time
International team takes important step towards better understanding the manifestations of the hereditary disease
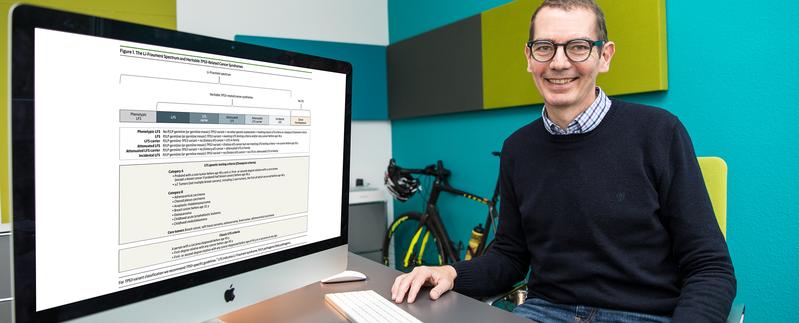
Li-Fraumeni syndrome (LFS) is associated with a dramatically increased lifelong risk of a broad spectrum of cancers and is caused by pathogenic variants in the TP53 gene. A definition reflecting the broad disease spectrum has been lacking and factors leading to differences in disease severity remain largely unknown.
An international research collaboration between Professor Christian Peter Kratz, Hannover Medical School (MHH), Germany, Professor Pierre Hainaut, Institute for Advanced Biosciences, Grenoble, France, Dr. David Malkin, The Hospital for Sick Children (SickKids), Toronto, Canada and other researchers has led to the development of a new LFS classification under the term "Li-Fraumeni spectrum" that comprehensively reflects the disease severity and facilitates the search for factors that lead to different disease courses. The research results were published in the renowned journal JAMA Oncology.
"A new classification was needed"
Clinical criteria can be used to identify people who are likely to carry a germline TP53 variant. However, more recently, it has become clear that individuals who do not meet these criteria can also harbor germline TP53 variants. The finding of an underlying TP53 pathogenic (disease-causing) variant is important because the diagnosis prompts cancer surveillance investigations that improve the prognosis of affected individuals. "The new classification was necessary to describe the evolving spectrum of LFS. It is based on established clinical criteria developed over decades and takes into account cancer risk-modifying factors without the need to specify them. At the same time, it can facilitate the search for factors that influence disease severity in people with LFS," says Professor Kratz, Director of the Department of Paediatric Haematology and Oncology of MHH.
3,034 patients from 1,282 families analysed
The research team used the classification to analyze data from 3,034 patients from 1,282 families with a hereditary TP53 variant from the International Agency for Research on Cancer database, identifying differences between TP53 variants in patients with severe disease compared to those with milder courses.
Differences in tumor characteristics
Of the 3,034 people, 2,139 (70.5 per cent) met the LFS genetic test criteria or developed cancer before the age 18 years. 149 people (4.9 per cent) were unaffected carriers and blood relatives of these patients. 678 persons (22.3 per cent) did not meet the LFS genetic test criteria and did not develop cancer before the age 18 years. 33 people (1.1 per cent) were cancer-free carriers and relatives of these patients. While the lifetime risk was about the same for patients who did or did not meet the LFS genetic cancer criteria, differences were seen in tumor patterns. "Patients who met the LFS genetic testing criteria had a higher incidence of early adrenal, brain, connective tissue and bone tumors, whereas TP53 variant carriers who did not meet the genetic testing criteria had a higher incidence of breast and other cancers, 45 per cent of which occurred after the age of 45," Professor Hainaut of Institute for Advanced Biosciences, Grenoble, points out.
The new LFS classification is a step towards better understanding the factors that lead to different manifestations. “The results of this study provide a rational basis on which to more accurately counsel patients with TP53 variants as to their own cancer risk, their family members’ cancer risk, as well as to more precisely utilize strategies for early tumor detection,” says Malkin of The Hospital for Sick Children, Toronto. “The approach we took lends itself to consider similarly re-classifying other cancer predisposition conditions.”
In addition to Germany, France, and Canada, researchers from Brazil, England, and the USA, were involved in the project. The study was funded in part by the German Childhood Cancer Foundation, by the Federal Ministry of Education and Research and by the Canadian Institutes of Health Research and the Terry Fox Research Institute.
Wissenschaftlicher Ansprechpartner:
For further information, please contact Professor Dr. Christian Kratz, telephone + 49 (511) 532-6712, kratz.christian@mh-hannover.de, or Professor Dr. Pierre Hainaut, telephone + 33 (0)6 20 38 05 47, pierre.hainaut@univ-grenoble-alpes.fr, or Dr. David Malkin, Dr. David Malkin, public.affairs@sickkids.ca.
Originalpublikation:
The original paper can be found here: https://jamanetwork.com/journals/jamaoncology/fullarticle/2785678?resultClick=1
Weitere Informationen:
http://Further information on the LFS can be found at https://www.lfsassociation.org.
Die semantisch ähnlichsten Pressemitteilungen im idw