When synapse building blocks became scarce: Bayreuth biologists explain protein exchange during vertebrate evolution
The electrical synapses in vertebrates are made of different, but by no means more powerful proteins than the electrical synapses in far older invertebrates. Animal physiologists at the University of Bayreuth have now found an explanation for this evolutionary puzzle. In the early phase of vertebrate evolution, there was a loss of diversity in precisely those proteins that had been used for signal transmission in older invertebrates. The scientists have published their discovery in the journal eLife.
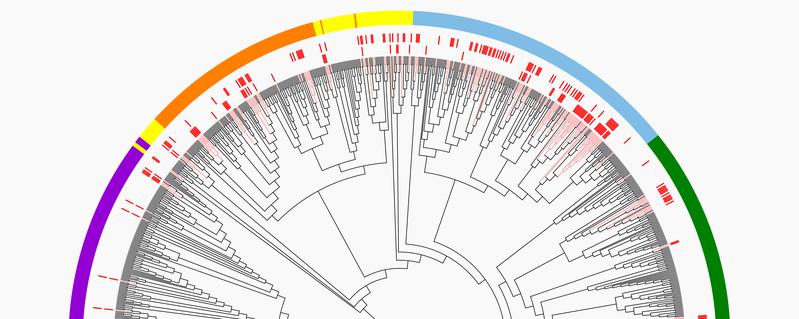
Electrical synapses establish connections between neighbouring cells and are of central importance for signal transmission in the body. Before the evolution of vertebrates, they were formed using innexins, a family of proteins with very diverse functions. For example, synapses consisting of innexins take on important functions in the nerve circuits in the brains of insects. In vertebrates, however, it was long assumed that electrical synapses do not consist of innexins, but exclusively of connexins. The new study by Bayreuth animal physiologists Dr. Georg Welzel and Prof. Dr. Stefan Schuster now provides the first comprehensive scientific confirmation of this assumption. There are, in fact, no invertebrates whose electrical synapses contain connexins. And conversely, no electrical synapses are found in vertebrates that consist of innexins, as used in invertebrates for signal transmission.
This finding seems puzzling because the synapses consisting of connexins do not give vertebrates any evolutionary advantage. In terms of their functions in signal transmission, they are not superior to the synapses based on innexins. Moreover, vertebrates do produce innexins, but not for use in electrical synapses. There must therefore have been another reason why the building blocks of the synapses were exchanged in the course of vertebrate evolution. The Bayreuth scientists have now discovered the explanation. In the early phase of vertebrate evolution, the diversity of innexins declined dramatically. Only one single innexin remained, which was unsuitable for use in synapses and had to take over other functions in the vertebrate organism – for example, the release of signalling molecules. It was not any superior functionality of connexins, but a lack of innexins that led to a complete replacement of synapse proteins in the course of vertebrate evolution. "Our findings suggest that a so-called genetic bottleneck at the beginning of vertebrate evolution led to this massive loss of innexin diversity," says Dr. Georg Welzel.
Even in the older invertebrates, as the study also shows, innexins have important functions beyond electrical synapses. In vertebrates, three highly conserved forms have evolved from the remaining innexin, but they occur as glycosylated proteins. This means that they form a chemical bond with carbohydrates, making them unsuitable as building blocks for synapses. Moreover, they are all extremely conserved during evolution. "The continued existence of innexins in vertebrate organisms and their extreme conservation during evolution suggest that they have very different but important functions. Despite their presumably great importance, these functions are still unknown and it will be important to identify them in the future," says Prof. Dr. Stefan Schuster.
Wissenschaftlicher Ansprechpartner:
Prof. Dr. Stefan Schuster
Animal Physiology
University of Bayreuth
Phone: +49 (0)921 55-2470
E-Mail: stefan.schuster@uni-bayreuth.de
Originalpublikation:
Georg Welzel, Stefan Schuster: Connexins evolved after early chordates lost innexin diversity. eLife (2022) DOI: https://doi.org/10.7554/eLife.74422
Die semantisch ähnlichsten Pressemitteilungen im idw