Plants use their roots to measure manganese concentration available in the soil
A team of researchers led by Jörg Kudla from the Institute of Biology and Biotechnology at the University of Münster are the first to demonstrate, using the model plant thale cress (Arabidopsis thaliana), how plants perceive manganese deficiency and which processes subsequently take place in the plant at the molecular level.
Every living organism needs the element manganese as an essential nutrient. In plants, for example, it plays a major role in breaking down water into oxygen and hydrogen during photosynthesis. A team of German and Chinese researchers are the first to demonstrate, using the model species thale cress (Arabidopsis thaliana), how plants sense manganese deficiency and which processes then take place in the plant at the molecular level. The researchers showed that a hitherto undetected group of cells in the plant root plays a decisive role. The researchers hope that the results of their work will in the future lead to methods for making plants more resistant to manganese deficiency – a condition which often occurs in alkaline and calcareous soils.
Prof. Jörg Kudla from the Institute of Plant Biology and Biotechnology at the University of Münster is one of the leading authors of the study and, as he says, “There have been a lot of studies focusing on which proteins are involved in the uptake and transportation of manganese within a cell. But how the manganese balance is regulated at the level of the organism is completely unknown.” Because calcium is involved as a messenger substance in numerous other regulating processes in the plant, the researchers asked themselves whether it also plays a role in regulating the manganese balance.
Manganese deficiency triggers oscillating calcium signals
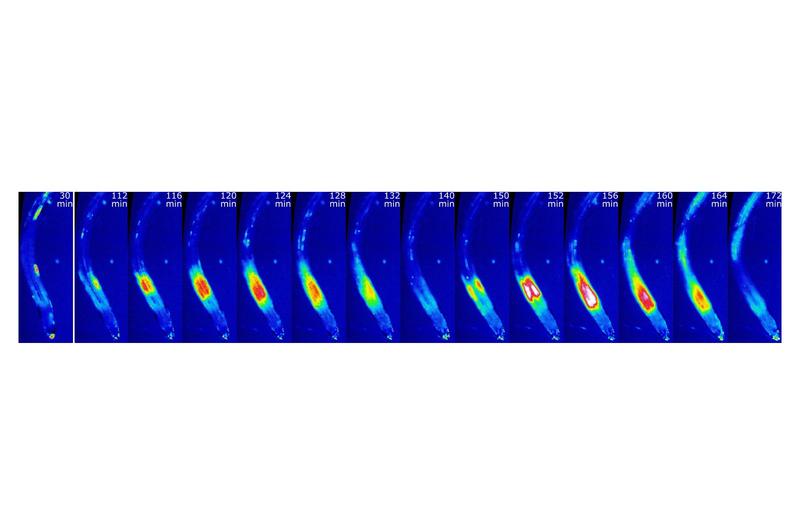
The team identified a special cell group in the plant root and gave it the name “manganese-sensitive niche”. Unlike all other root cells, these cells display a special reaction in response to manganese deficiency: the calcium concentration within the cells rises and falls, several times in succession, as long as the deficiency lasts. Every oscillation lasts approximately 30 minutes. “Nobody had previously observed such multi-cellular oscillations in the calcium concentration which are built up through the coordinated occurrence of calcium signals in individual cells in plants,” says Kudla. Just a few hundred cells jointly build up the signal. The epidermal cells – the cells in the outermost root layer – are the first to begin increasing the calcium concentration. Then the cells situated further inside gradually follow suit – before the whole process is then reversed.
Third stimulus-specific “sensitive niche” now discovered
In earlier work, researchers led by Jörg Kudla had already discovered two further “sensitive niches” in other areas in the root – a potassium-sensitive niche and a sodium-sensitive niche. Here, too, the roots reacted by producing multicellular calcium signals in specific cell groups in response to changes in the ion concentrations in the environment. However, the researchers did not observe any oscillations – unlike in the manganese-sensitive niche.
In their current study, the researchers discovered that the calcium oscillations triggered by manganese deficiency activate two special enzymes – so-called Calcium-Dependent Protein Kinases (CPK21 and CPK23) – and that these two enzymes, for their part, stimulate the uptake of manganese. “As the kinase frees itself from the calcium, and these again become inactive. Our hypothesis is that every oscillation starts this process anew – until the plant has achieved a sufficient uptake of manganese,” says Kudla. The manganese transporter NRAMP1, which is responsible for transporting the manganese into the cells of the root, is part of the process. The protein kinases CPK21 and CPK23 interact with this transporter and regulate the uptake of manganese by phosphorylating one specifc amino-acid (Thr498).
In order to demonstrate the occurrence of the calcium signals, the researchers used high-resolution microscopy and, for the first time, ultra-sensitive molecular calcium-biosensors. Biosensors generally visualise changes in concentrations of bioactive substances such as calcium in cells and tissues. The team combined these studies, involving “in vivo biosensor technology”, with genetic, cell-biological and biochemical methods in order to clarify the underlying molecular mechanisms. In addition to the Münster researchers, scientists from the College of Life Sciences, Northwest A&F University in Shaanxi and the Biotechnology Research Institute, Chinese Academy of Agricultural Sciences in Beijing (China) as well as from the Martin Luther University Halle-Wittenberg were also involved.
Funding:
The work received financial support from the National Natural Science Foundation of China (NSFC) and from the German Research Foundation (DFG).
Detailed image line:
Arabidopsis seedlings have been exposed to manganese deficiency, and cytosolic calcium dynamics have been visualized utilizing the GCaMP6f-mCherry calcium biosensor. The calcium dynamics in roots are presented as false color images for selected timepoints after onset of manganese deficiency (from left).
Wissenschaftlicher Ansprechpartner:
Prof. Jörg Kudla
Institute of Plant Biology and Biotechnology
University of Münster
Phone: +49 251 83-23808
Mail: jkudla@uni-muenster.de
Originalpublikation:
Dali Fu et al. (2022): Ca2+-dependent phosphorylation of NRAMP1 by CPK21 and CPK23 facilitates manganese uptake and homeostasis in Arabidopsis. Proc Natl Acad Sci USA 119(40); https://www.pnas.org/doi/10.1073/pnas.2204574119. Epub 2022 Sep 26.
Weitere Informationen:
https://video.uni-muenster.de/de/mp4/1-140%2010Fps.mp4 Video: Cytosolic calcium dynamics after onset of manganese deficiency (Copyright: WWU - AG Kudla)
Ähnliche Pressemitteilungen im idw