Researching how pepper grows
Leibniz Institute of Plant Biochemistry sheds light on the biosynthesis of pungency
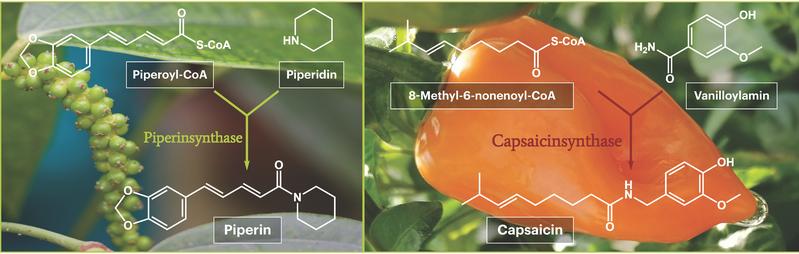
How do plants make those pungent substances, anyway? The Leibniz Institute of Plant Biochemistry (IPB) is working intensively on this topic. Recently, a group of scientists led by Dr. Thomas Vogt have pinpointed the crucial enzyme that gives the fruits of the pepper plant (lat. Piper nigrum) the distinctive, pungent taste. The enzyme in question, piperine synthase, catalyzes the final step towards biosynthesizing pungent piperine. Now the biochemists have followed up with a second pungent compound by shedding more light on how capsaicin from chili peppers (Capsicum spec.) is biosynthesized.His research group for the first time describes the enzymatic activity of the long-sought capsaicin synthase. The enzyme is what catalyzes the final reaction step when a plant produces capsaicin.
When piperine and capsaicin come into contact with the tongue and mucous membranes, they bind to the exact receptor that also reacts to heat, acids or injury. This receptor then triggers a stimulus in certain nerve cells, which humans perceive as spiciness. Capsaicin and piperine also have antimicrobial, digestive and circulatory effects. So over and above spicing dishes up, they also hold considerable promise as active ingredients for medical applications. "Recent literature includes hundreds of studies on the biological effects of pepper and chili extracts, but very few cover how the pungent substances are actually biosynthesized," explains Thomas Vogt, who heads the Phenylpropane Metabolism research group at the IPB.
One reason may be the special combination of expertise required to elucidate the biosynthetic pathways of a plant, which is seldom in evidence, except for a few research institutes. Ideally, chemists, biochemists and bioinformaticians join forces to zoom in on the handful of candidate enzymes involved in producing a sought-after substance. Compounding the search for the enzymes is the fact that numerous genes and enzymes are still unknown in certain plant species. "I mean here," says Thomas Vogt, "you don't have much leeway to consult the databases."
When it came to pepper, as well as this lack of data, other issues also emerged. For example, the Halle scientists first had to grow pepper plants, then, come what may, ensure that they bore fruit. "That's not a given with pepper, which grows in a greenhouse," explains the biochemist, "but the gardeners at the Institute succeeded." Having successfully cultivated the plants, the research team could harvest pepper berries to various stages of maturity over a three-month period and closely observe how their piperine content steadily increased. According to the scientists, the ripening berries should also contain the enzymes that form piperine. The plant's leaves, conversely, should not contain the biosynthesized piperine enzymes, because they do not produce piperine and therefore do not taste spicy.
The scientists exploited these differences in piperine content, i.e. when the piperine biosynthesis enzymes were present and absent respectively. Taking the premise that an enzyme is present in certain tissues only when its gene is activated and read, they compared gene activities in leaves and fruits at different stages of maturity (ripeness). Doing so paved the way for them to identify those genes that were particularly active in immature fruit, including the specific gene for piperine synthase. This gene was then introduced into microorganisms, where it served as a template for the bacteria to produce the piperine synthase enzyme. All of which allowed the pepper experts from Halle to prove beyond doubt that the isolated enzyme catalyzes the fusion of the two starting substances piperoyl-CoA and piperidine to form piperine. Confirmation of the long-sought-after piperine synthase was thus achieved.
But the task of discovering capsaicin synthase brought other problems with it. Here, although the coding gene had already been identified, no scientist had ever successfully produced an enzymatically active protein from it in bacteria. In other words, the cast-iron proof that the gene under investigation was capsaicin synthase remained pending. Despite this, the plant researchers from Halle leveraged their expertise while working on the relatively similar pepper enzyme to isolate capsaicin synthase and prove in the final activity test that it did indeed catalyze the long-postulated reaction of the two starting substances, 8-methyl-6-nonenoyl-CoA and vanilloylamine to capsaicin. These final proofs of activity for piperine and capsaicin synthase were achieved partly because the Institute's synthesis chemists were able to produce all the starting materials themselves, despite them not yet being available for purchase, and use them as required during the enzyme tests.
With the main reaction steps of piperine biosynthesis and capsaicin biosynthesis now elucidated, the prospect of fully understanding both biosynthetic pathways at some point remains. But why is this finding a game-changer? Once you know all the enzymes involved in a biosynthesis process, you can introduce the appropriate genes into microorganisms and have them produce the desired active ingredient. "For pepper, however, it wouldn't make sense economically," says Thomas Vogt, "because piperine is present in pepper berries in such high concentrations that isolating it, e.g. for medicinal purposes, would be easy."
Even so, the IPB scientists find the task of elucidating biosynthetic pathways rewarding, since newly discovered enzymes catalyze interesting reactions that spawn reaction products with complicated structures. These enzymes can be modified by biochemists to create completely new enzymes with desired properties. Once optimized, these enzymes are then used to synthesize new and potentially effective substances in the test tube. This type of biocatalytic synthesis, namely mimicking and optimizing the original plant biosynthesis in the test tube, holds a wealth of potential going forward as a relatively young research field. As an alternative to petrochemical syntheses, biocatalysis can generate desired substances without requiring the use of any toxic catalysts and solvents and generating no harmful byproducts.
Background: Biosyntheses and enzymes
Enzymes are proteins that are prevalent in every living being and in every cell. Every reaction in the body in which a substance is converted into another substance (metabolism) is catalyzed by an enzyme. Every organism houses thousands of different metabolic reactions, involving just as many different enzymes. Several successive individual reactions form separate metabolic or biosynthetic pathways that lead to many different end products. In terms of ability to produce complex and wide-ranging substances, plants outperform animals. That is also why their enzyme count far outweighs that of animal organisms. Already for millennia, people have been using enzymes to produce bread, sauerkraut, alcohol and cheese to name but a few.
Wissenschaftlicher Ansprechpartner:
Dr. Thomas Vogt
Leibniz Institute of Plant Biochemistry
thomas.vogt@ipb-halle.de
Originalpublikation:
Arianne Schnabel, Benedikt Athmer, Kerstin Manke, Frank Schumacher, Fernando Cotinguiba & Thomas Vogt. Identification and characterization of piperine synthase from black pepper, Piper nigrum L. Communications Biology 4, DOI: 10.1038/s42003-021-01967-9
Raika Milde, Arianne Schnabel, Toni Ditfe, Wolfgang Hoehenwarter, Carsten Proksch, Bernhard Westermann & Thomas Vogt. Chemical Synthesis of Trans 8-Methyl-6-Nonenoyl-CoA and Functional Expression Unravel Capsaicin Synthase Activity Encoded by the Pun1 Locus. Molecules 27 (20), 6878; https://doi.org/10.3390/molecules27206878
Weitere Informationen:
https://www.ipb-halle.de/en/public-relations/press-releases/press-detail/forschen-wie-der-pfeffer-waechst-ipb-klaert-biosynthese-der-schaerfe-auf/
Ähnliche Pressemitteilungen im idw