New study reveals ‘moonlighting’ function of part of the brain’s protein destruction machine at synapses
A major component of the cell’s protein destruction machine moonlights at brain synapses
A new study by researchers at the Max Planck Institute for Brain Research discovered a ‘moonlighting’ function carried out by a complex that normally works to degrade proteins in cells - this protein destruction machine is called the proteasome. The scientists found, by counting and visualizing individual protein complexes, that one part of the proteasome (the 19S regulatory complex) was abundant near brain synapses where it regulates synaptic proteins and transmission on its own - without its partner.
The human brain possesses over 100 trillion synapses. These synaptic connections define neural circuits and store information for as long as lifetime. However, the major functional molecules inside the synapses, the proteins, only have an average ‘shelf-life’ of one week. This means that synapses need to constantly degrade old proteins and synthesize new copies to replace them. The homeostasis of proteins at synapses is fundamental to all aspects of brain function. And its imbalance leads to neurological disorders. Most of the brain’s cellular proteins are degraded by a multi-component machine called the proteasome.
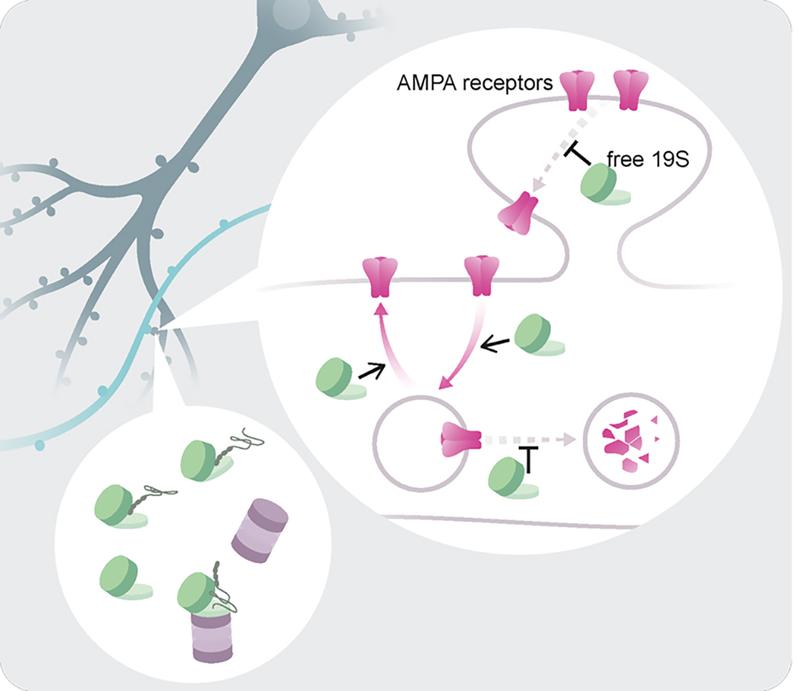
The proteasome, the cell’s protein garbage disposal system, has two parts: one for recognizing the proteins (the so-called 19S complex) and the other for breaking them down (the 20S complex). Previously, it was thought that the 19S only worked together with its partner, the 20S complex. A recent study in Science from Professor Erin Schuman’s lab (also Director of the Max Planck Institute for Brain Research) shows that this is not always the case. In the remote regions of the neuron- the dendrites, where information is transferred from one neuron to another at synapses- “super-resolution” visualization methods showed that the 19S complex of the proteasome is much more abundant than the 20S and can often be found alone. Surprisingly, functional studies showed that the free 19S particle regulates synaptic proteins without promoting their degradation. It recognizes a special group of synaptic molecules (e.g. neurotransmitter receptors, also called AMPA receptors) and changes their dynamics and localization within the synapse. This study reveals an intriguing and flexible aspect of synapse biology, where complex protein machines have likely adapted to subcellular needs and take on alternative functions.
The work received generous support from many members of the Schuman lab, the imaging facility and the proteomics facility at MPIBR. The lead author, Dr. Chao Sun, is now an independent group leader at the Danish Research Institute of Translational Neuroscience – DANDRITE (Nordic-EMBL Partnership for Molecular Medicine) and Aarhus University, Denmark.
Wissenschaftlicher Ansprechpartner:
Chao Sun , Ph.D.
Institut for Molekylærbiologi og Genetik - Neurobiologi
Universitetsbyen 81
8000 Aarhus C
Dänemark
chaosun@dandrite.au.dk
Prof. Dr. Erin M. Schuman
Max Planck Institute for Brain Research
Max-von-Laue-Str. 4
60438 Frankfurt am Main
erin.schuman@brain.mpg.de
Originalpublikation:
DOI: 10.1126/science.adf2018
Die semantisch ähnlichsten Pressemitteilungen im idw