Learning from viruses: Molecular fibers can help to introduce genetic material into cells
Many viruses manage to overcome the body's own barriers and enter human cells. This function could be useful in the future for so-called "gene therapy." Inactivated viruses can be "programmed" with new functions to transport genetic material into cells. Researchers led by Tanja Weil and Christopher Synatschke of the Max Planck Institute for Polymer Research, in close collaboration with Jan Münch (University of Ulm), have now studied in detail long molecular fibers consisting of so-called peptides that enhance the infection process of viruses.
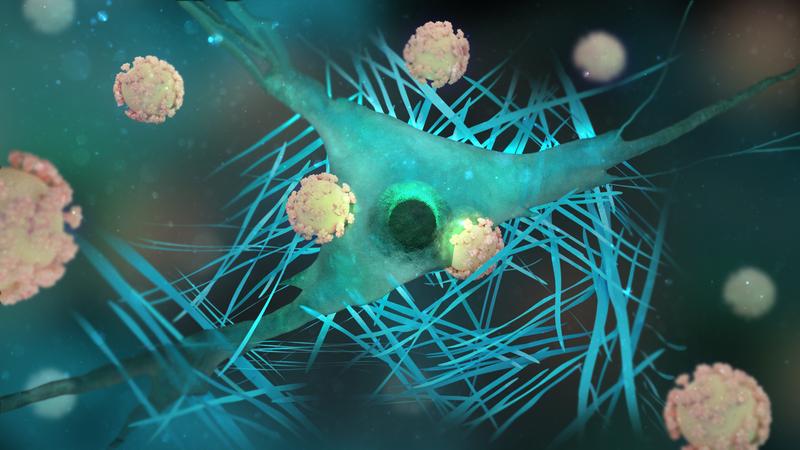
Pathogenic viruses that enter the human body can dock onto cells with their tentacle-like extensions, whereupon the cell takes up the viruses. This process, which is already known and occurs in diseases such as HIV, can also be used for therapeutic approaches.
In so-called "gene therapy", the aim is to introduce genetic material into cells. This allows a variety of applications: Defective genes in cells, such as those found in hereditary diseases, can be specifically replaced to restore the missing cell functionality. Another example are T cells, immune cells of the body, which can be reprogrammed to attack emerging cancer. In all cases, efficient transport of genetic material into the cell is necessary.
Research is now exploiting this ability of viruses to enter cells. The protein extensions that are docked onto the viral shell are an important building block for this: they ensure that the viral shells dock onto the cell.
Researchers led by director Tanja Weil and group leader Christopher Synatschke have now taken a closer look at a small fragment of these protein extensions on different size scales. The molecule - a so-called "peptide" - consists of a chemical compound of different amino acids. Several of the molecules joined together can form long, spaghetti-like structures called "peptide fibers." These fibers can serve as a kind of "glue" between so-called therapeutic viruses and cell envelopes, improving the process of virus uptake into cells. Improved binding can reduce the viral dose needed for gene therapy and increase the chances of successful therapy.
The team has now investigated how the molecular composition of different amino acids influences the structure-forming properties: in other words, how is the formation of fibers or the binding between virus and cell influenced by the molecular structure?
To this end, they have studied over 150 different molecules based on the original protein fragment. To arrive at this variety of molecules, they systematically replaced individual amino acids or swapped them within the molecule.
"Some of the more than 150 peptides studied indeed caused the binding of the viral shell to the cell to be enhanced. In others, no fiber aggregates formed at all that could have provided binding," said Kübra Kaygisiz, first author of the publication. "We have now tried to find out which chemical properties of the molecules are primarily responsible for the binding," adds Christopher Synatschke.
"We were able to use computational methods in our experimental study to identify different properties that enable efficient gene transfer," says Kübra Kaygisiz. "Surprisingly, our molecules also need to behave in a water-repellent - hydrophobic - manner as well as have an alternating arrangement of hydrophobic with positively charged groups within the molecule to increase virus binding," explains Kübra Kaygisiz. They were also able to find this principle in other naturally occurring protein fragments, suggesting a universal property-activity relationship that enabled artificial intelligence to predict new fiber materials.
Their findings could make gene therapy more effective in the future: Less drug, due to greater uptake by the cell, could significantly improve the chances of therapy. They have published their results in the renowned journals "Nature Communications" and "Biomaterials Science".
Wissenschaftlicher Ansprechpartner:
Dr. Christopher Synatschke
synatschke@mpip-mainz.mpg.de
Originalpublikation:
Kaygisiz, K.; Rauch-Wirth, L.; Dutta, A.; Yu, X.; Nagata, Y.; Bereau, T.; Münch, J.; Synatschke, C.; Weil, T.: Data-mining unveils structure–property–activity correlation of viral infectivity enhancing self-assembling peptides. Nature Communications 14, 5121 (2023).
DOI: /10.1038/s41467-023-40663-6
Ähnliche Pressemitteilungen im idw